RESEARCH
An essential function of the brain reward system is to determine whether something is good or bad, which is critical to an organism’s survival and reproduction. The reward pathway has evolved to ensure that meaningful stimuli for animals, such as food, sex, and social interactions are perceived as rewarding. Although the mechanisms of sensory detection of these stimuli have been intensively studied, it remains mysterious how the detected sensory signals are transformed into the perception of reward or valence.
There are usually multiple layers of neurons between peripheral sensory receptors and reward neurons in the central brain. These interconnected intermediate layers are where multisensory integration takes place. It is common in the natural environment that hungry animals hesitate about pursuing food and mates when a predator suddenly appears. The simultaneous valuation of multimodal sensory stimuli is critical to higher cognitive functions, such as decision making and learning. Therefore, these intertwined and undescribed intermediate layers are key to the proper tuning of reward perception by internal states and external stressors.
Resolving this knowledge gap will require delineating the anatomy and functional structure of the neural pathways that mediate the transformation of sensory stimuli to reward perception. We will address this question in Drosophila melanogaster, a tractable system with simple brain circuits and an extensive genetic toolbox for neurogenetic and neurocircuitry studies. Our goal is to define the neural architecture and principles by which reward information from different sensory modalities is perceived and processed. To this end, we will work on the following projects.
- Delineating the neural pathways that mediate different rewarding stimuli, such as food and sex.
- Determining how perceived rewarding stimuli are integrated and interact with each other.
- Probing how this process is tuned by internal states and environmental stresses.
Why flies?
The brain reward system is very complex, consisting of millions of neurons. This figure shows a comparison of the sizes of a human, a mouse, and a fly brain. The brain of the fruit fly, Drosophila melanogaster, has only about 100,000 neurons, about 200 of which are dopaminergic neurons. Although considered as very simple creatures, flies have enriched behavior reservoirs. Besides, there are numerous genetic resources and toolkits for fly research.
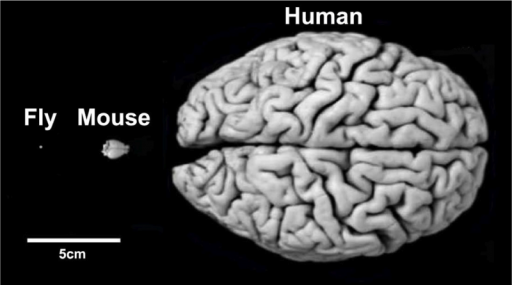
Despite their differences in size and appearance, the brains of flies, mice and humans rely on similar structures regulating comparable behaviors under similar principles. (Photo: Frank Hirth/KCL)
What approaches?
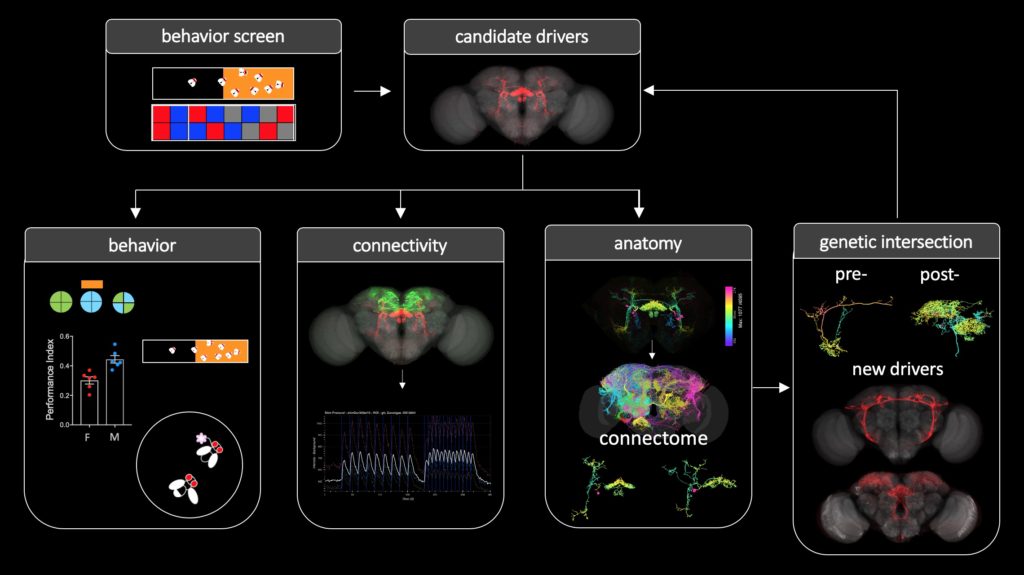
We adopt and combine research approaches at different levels to pursue our goals, including molecular, connectome, imaging, and behavioral analysis. This figure illustrates a general workflow.